 Installation Qualification should be requalified if: A component is assembled against the recommendation of the manufacturer.Utility connection in accordance with applicable specifications.List of Instruments that require qualification.A review of all design features, such as dimensions, make, model, materials, etc., in relation to the Design Qualification specification and/or the manufacturer’s claim.Compliance with all specifications described in the Design Qualification and/or manufacturer’s specification.The installation qualification test includes checking: Specifications of the equipment received from the Manufacturer / Vendor at the site of the User.Verification of the design of the equipment.SAT is conducted to ensure that the instrument or equipment’s physical condition and availability of all components. Ensure that faulty components can be modified, adjusted, and replaced without having to rely on fabrication shops.Prior to shipping, make sure all failures out of the box have been eliminated.Confidence that the equipment/instrument/system will perform effectively under the full range of possible circumstances.The equipment/instrument/system must function properly, have high quality, and be of high integrity.Make sure the equipment/instrument/system is functioning.Document quality (availability) verification.ĭuring the Factory Acceptance Test, the following should be accomplished:.Using conventional function testing or simulation, the functionality of the product is determined.Verification of URS against the actual.Tests the completeness of the installation.FAT shall be conducted at the vendor manufacturing site. Factory Acceptance Test (FAT):įAT has some important checkpoints that shall be verify while this FAT test. 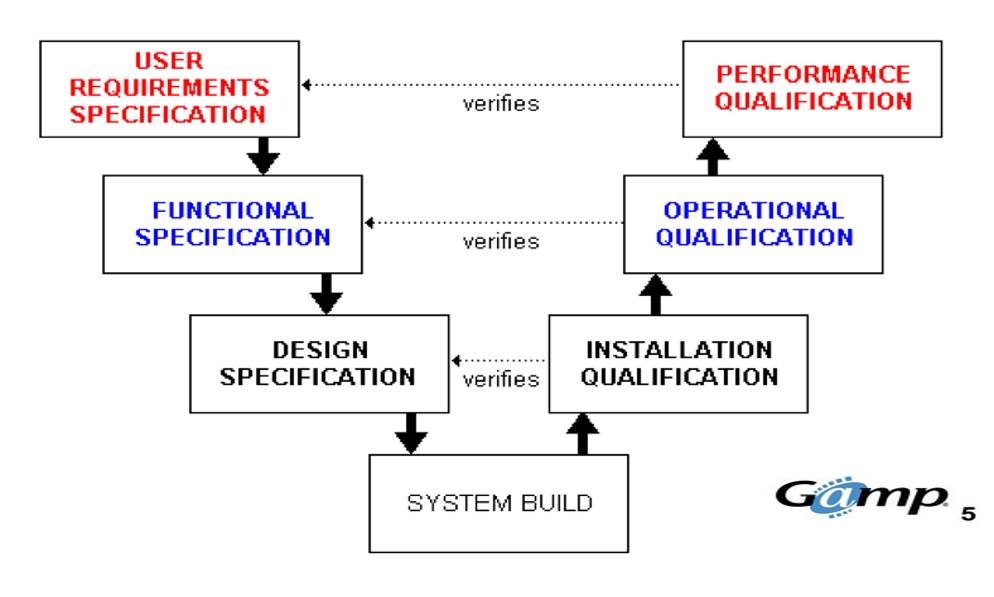
The Design Qualification is an activity that is part of the documentation and recommended to be completed before placing the purchase order. Specifications that specify the design and functionality of equipment, verified with the actual equipment details provided by our suppliers such as design and functionality. of equipment, so they can be used to specify their design. In a design specification, the details of how the equipment must be designed, e.g. Specifically, Design Specifications provide knowledge of dimensions, construction materials, layouts, etc. User Requirement Qualification (URS):ĭesign and Functional Specifications are part of user requirements specifications. Let’s understand the steps of qualification. 
Site Acceptance Test (SAT) at the user site.Factory Acceptance Test (FAT) at the vendor site.User Requirement Specification is well known as “URS”.To know the basics of equipment qualification, important to know what are the steps involved in qualification? 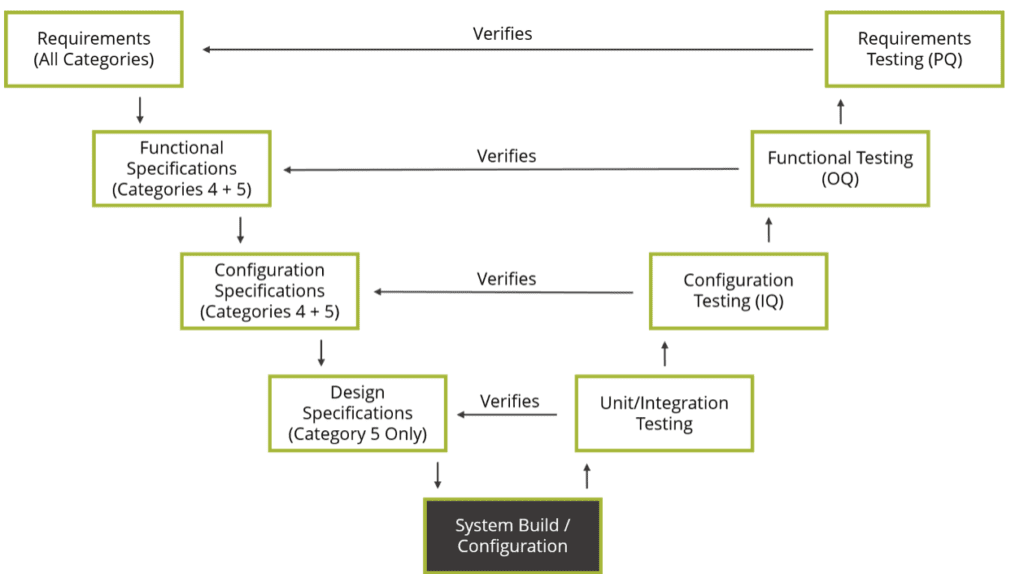
What are the steps involved in equipment qualification? What is a need for Equipment qualification? First, need to understand what is a need for equipment qualification. What are the steps involved in equipment qualification?Įquipment Qualification Principles And PracticesĮquipment qualification is an important process for any equipment or instrument used in the pharmaceutical industry.What is a need for Equipment qualification?.Equipment Qualification Principles And Practices. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
